|
10/2/2023 0 Comments Nutrfs
Most patients were White (75% vs 76%), had an ECOG performance status of 0 (63% vs 62%), and had a tumor originating in the urinary bladder (79% vs 79%). The median age in the nivolumab and placebo groups, respectively, were 65.3 years (range, 30-92) vs 65.9 years (range, 42-88). Exploratory end points included distant metastases-free survival (DMFS), second progression-free survival (PFS2), safety, and health-related quality of life. Secondary end points included NUTRFS, DSS, and overall survival. The primary end point was DFS in the ITT and PD-L1 of 1% or more populations. Stratification factors included tumor PD-L1 status, previous neoadjuvant cisplatin-based chemotherapy, and nodal status. Patients were randomly assigned 1:1 to receive intravenous nivolumab at 240 mg every 2 weeks or placebo followed by adjuvant treatment for up to 1 year. 
Eligible patients also underwent radical surgery within the last 120 days and attained disease-free status within 4 weeks of randomization. The randomized, double-blind, multi-center study included patients with ypT2 to ypT4a or ypN-positive disease who had previously received treatment with neoadjuvant cisplatin. Nivolumab was favored over placebo across all prespecified patient subgroups, including those based on age, sex, region, performance status, minor histologic variance, pathologic lymph node status, and pathological T stage, Galsky noted. “These results further support adjuvant nivolumab as a standard of care for patients with muscle-invasive urothelial cancer at high risk for recurrence after radical surgery,” according to lead author, Matthew Galsky, MD, a professor of medicine, director of genitourinary medical oncology, and co-director of the Center of Excellence for Bladder Cancer at the Tisch Cancer Institute, as well as the associate director for translational research at the Tisch Cancer Institute, Mount Sinai in New York, New York. In the population with a PD-L1 expression of at least 1%, the rates were 60.3% vs 37.6% and 56.9% vs 33.3%, respectively. The 24-month and 36-month DFS rates for the nivolumab and placebo arms, respectively, were 48.4% vs 38.8% and 45.0% vs 34.9% in the ITT population. In the population with a PD-L1 expression of 1% or more, the median DFS was 52.6 months (95% CI, 25.8-not estimable ) and 8.4 months (95% CI, 5.6-17.9), respectively (HR, 0.52 95% CI, 0.37-0.72). In the intent-to-treat (ITT) population at a median follow-up of 36.1 months vs 33.9 months, respectively, nivolumab continued to yield DFS improvements vs placebo, including in patients with a PD-L1 expression of 1% or more. The findings, presented at the 2023 Genitourinary Cancer Symposium, support nivolumab’s role as standard of care therapy in this setting, according to investigators. 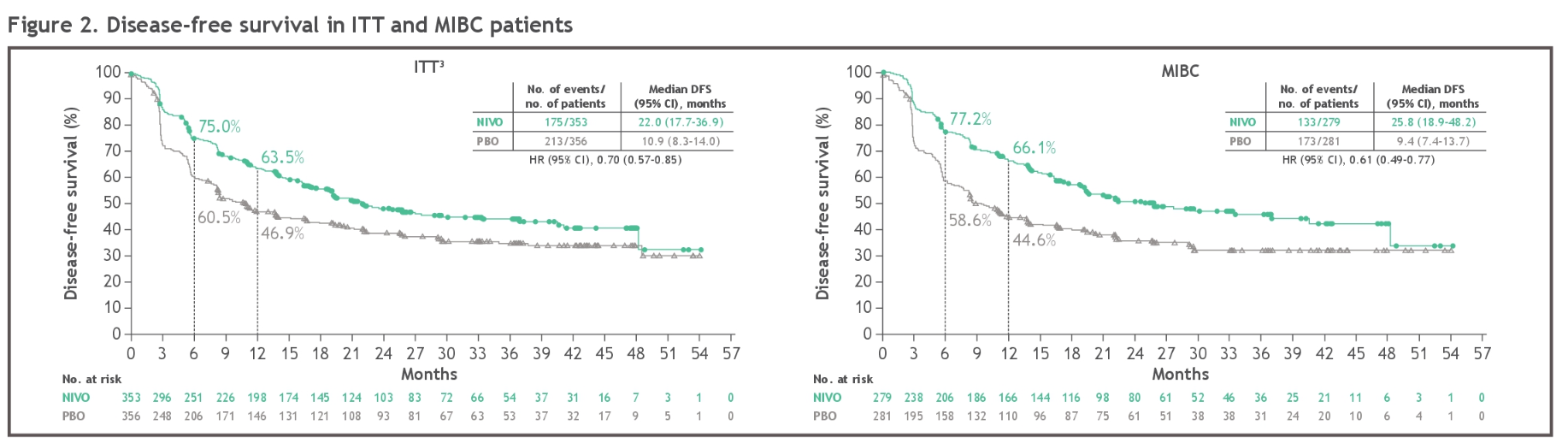
Adjuvant treatment with nivolumab (Opdivo) yielded better disease-free survival (DFS), non-urothelial tract recurrence-free survival (NUTRFS), and disease-specific survival (DSS) rates than placebo in patients with high-risk muscle-invasive urothelial carcinoma who had undergone radical surgery, according to a long-term follow-up from the phase 3 CheckMate 274 study (NCT02632409).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
